- Nick Water DR
- Published
- Updated February 27, 2026
- Articles
Chlorine in our water and what about the byproducts !
127 Years of Chlorine — And It Never Stops
In 1897 a typhoid outbreak hit Maidstone, Kent. A bleach solution was pumped into the water main. It worked — and Britain had invented mass water chlorination. By 1905, Lincoln’s water supply was being chlorinated permanently, the first continuous programme in the world. The country that invented this system now has the weakest regulatory protections against its chemical byproducts in the developed world.
Chlorine is not occasional. It is dosed automatically, 24 hours a day, 365 days a year. Although there is no formal limit like in the US , treatment works add 0.5–1.0 mg/L by your tap it has dissipated to 0.1–0.8 mg/L. Online analysers adjust the dose in real time. Booster stations top up the residual across long networks. Without it, bacteria would colonise the pipes before water reached your home. Chlorine kills all sorts of bacteria E. coli, Salmonella, Typhoid, Legionella, and many viruses. It does not remove many of the more modern 21 st century contamination problems like heavy metals, nitrates, pharmaceuticals, PFAS , pesticides or microplastics.
The Chemistry Nobody Puts on the Label
Chlorine reacts with naturally occurring organic matter (NOM) — dissolved plant decay, soil compounds, fulvic acids — still present in water after treatment. Conventional coagulation and filtration removes 40–60% of these organic compounds but the low-molecular-weight fraction most reactive with chlorine is the hardest to eliminate. What remains becomes the raw material for disinfection byproducts (DBPs).
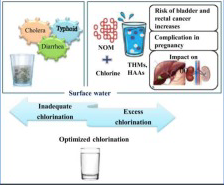
THMs (Trihalomethanes) — chloroform, BDCM and others. Volatile. A 2025 Swedish analysis found THM exposure may increase bladder cancer risk by 33% and colorectal cancer by 15% even at levels below legal limits.
HAAs (Haloacetic Acids) — DCA, TCA and others. Non-volatile; primary risk is ingestion. Six HAAs are classified by the US National Toxicology Programme as ‘reasonably anticipated human carcinogens.’ California’s safety threshold for some HAAs is 0.03 µg/L — over 2,000 times lower than the US legal limit of 60 µg/L.
Chloramines — formed when chlorine reacts with ammonia, sometimes added deliberately as a secondary disinfectant. More stable than chlorine, travels further in pipes. Its own byproducts include NDMA (a potent probable carcinogen) and iodoacetic acid. Standard carbon filters remove chloramines poorly.
Carcinogen Classifications

How DBPs Enter Your Body: The Skin Route
Dermal absorption of chemicals from the shower have been consistently underestimated , chemicals that are fat-soluble like THMs pass through it readily + regulatory safe levels for water contaminants have historically been calculated primarily on ingestion .Ingestion during a shower through the skin and from volatile compounds in the steam can meaningfully increase the intake of chemicals particularly in poorly ventilated showers . The dermal dose from a 10-minute shower is approximately equal to the dose from drinking 2 litres of the same water .
Its also notable that inhaling aerosolized chloramine or other chemicals from steam in your shower can lead to negative health issues.

Temperature amplifies this significantly. Research in Environmental Health Perspectives found dermal flux of DBPs increases 50–170% as water rises from 25°C to 40°C — the typical range of a hot shower or bath. Haloketone permeability triples at bathing temperature. Common surfactants in soap and shampoo (SLS, SLES) further increase skin permeability to chlorinated compounds by disrupting the stratum corneum lipid layer.
Not all DBPs cross skin equally .

Swimming pools concentrate this further: pool DBP levels run 10–100x higher than tap water, with over 110 individual DBPs identified. Exposure occurs via five simultaneous routes — dermal, inhalation, ingestion, buccal, and aural. A bladder cancer case-control study found higher odds ratios for showering and bathing exposure than for ingestion alone, meaning filtering drinking water while showering in unfiltered water addresses only part of the problem.
Children face higher proportional exposure — greater surface area to body mass ratio means a higher dermal dose per kilogram at any given DBP concentration
When Home and Office Conditions Make It Worse
DBP formation does not stop at the treatment works. Residual chlorine continues reacting with residual NOM throughout the distribution network and inside your own premises. Studies consistently show DBP concentrations increase between works and tap — particularly where water is warm, slow-moving, or pipe materials introduce additional organic nitrogen.

The first draw problem: Stagnant water in household pipes overnight has the highest DBP concentration of any draw from that tap. Run the cold tap for 20–30 seconds before drinking — especially first thing in the morning or in offices with low-use pipe runs.
Never use hot tap water for drinking , cooking, or infant formula ! The heat both increases DBP formation and is the primary advice given for lead leaching, with DBP concentration an equally valid reason.
What Actually Filters It Out
Most UK filters carry only NSF 42 certification — chlorine taste and odour. Not DBPs. There is no NSF certification specifically for HAAs, meaning no filter can be independently verified for HAA removal, even where the technology (reverse osmosis, anion exchange) is effective in practice . A whole house reverse osmosis or catalytic carbon system at the mains entry point is the only solution addressing all exposure routes — drinking, cooking, showering, and bathing simultaneously. Everything else is a partial solution this is why it important to carefully plan solutions that are affordable and practically sustainable !

The Bottom Line
Chlorine saves lives. Without it, waterborne disease would return to British towns within weeks. The science on that is unambiguous. What is equally unambiguous: the byproducts chlorine creates enter your body through multiple routes — not just what you drink, but what you absorb during every shower and bath you take. THMs cross skin readily. HAAs do not, but they remain in every glass you pour and every pot you boil. Neither are adequately regulated in the UK.
The US capped HAAs in 1998. The EU mandates a limit from 2026. California’s scientists calculate some HAAs become a cancer risk at concentrations 2,000 times lower than what US law allows. The UK has no legal HAA limit at all.
The water from your tap meets current legal requirements but this is not without risks and gaps that need to be considered and understood .
For a free consultation o this and all your water solutions BOOK HERE

About the Author
Nick Smith | Founder | The Water Dr. & Cellthyhomes
Nick has dedicated years to studying building biology, healthy living environments, and the impact of environmental toxins on inflammation.
Whilst regulations for UK drinking water are slow to adapt, & influenced by conflicts of interest, Nick conduct comprehensive research on global regulations & scientific literature to offer water filtration solutions that provide clean drinking water free from all harmful contaminants.