What limescale is really costing you, how each treatment works, and why one size does not fit all
Over 60% of UK homes roughly 17 million households ‚ are in hard water areas. For most of those households, the response to limescale is to buy descaler, scrub the showerhead, and otherwise ignore the problem. That is an expensive mistake. Hard water is not cosmetic. It is a progressive engineering and financial problem that compounds silently inside your boiler, pipes and appliances and it interacts with your health , your skin and your hair in ways the industry rarely explains clearly.
This guide covers everything: the chemistry of why hard water causes the problems it does, what limescale is actually costing you, how each treatment technology works (and where it fails), what UK law requires when you install one, and crucially ‚ why the right answer depends entirely on your water hardness, your priorities, and your household. One solution does not fit every home.
Part 1: The Problem — What Hard Water Does and Where It Does It
Hard water forms when rainwater filters through chalk and limestone geology. As it percolates through the rock, it picks up dissolved calcium (Ca²⁺) and magnesium (Mg²⁺) ions. By the time it reaches your tap, those ions are chemically stable — held in solution as calcium bicarbonate (Ca(HCO₃)₂), which is soluble, colourless, and at low-to-moderate concentrations, biologically beneficial.
The problem begins the moment you heat it.
The chemistry: When calcium bicarbonate is heated above approximately 60°C, the equilibrium breaks down: Ca(HCO₃)₂ → CaCO₃ (limescale) + H₂O + CO₂. The calcium carbonate that precipitates is insoluble. It bonds to whatever surface it contacts — heat exchanger, pipe wall, element — and stays there. The hotter the surface, the faster this happens. This is why boiler heat exchangers and immersion elements accumulate scale far faster than cold pipes. Limescale is not a stain. It is a thermodynamic insulator bonded to the surfaces your heating system depends on.
Hard water forms when rainwater filters through chalk and limestone geology. As it percolates through the rock, it picks up dissolved calcium (Ca²⁺) and magnesium (Mg²⁺) ions. By the time it reaches your tap, those ions are chemically stable — held in solution as calcium bicarbonate, which is soluble, colourless, and at low-to-moderate concentrations, biologically beneficial.
The problem begins the moment you heat it.
When calcium bicarbonate is heated above approximately 60°C, the equilibrium breaks down: calcium bicarbonate converts to calcium carbonate (limescale), water, and carbon dioxide. The calcium carbonate that precipitates is insoluble. It bonds to whatever surface it contacts — heat exchanger, pipe wall, element — and stays there. The hotter the surface, the faster this happens. This is why boiler heat exchangers and immersion elements accumulate scale far faster than cold pipes. Limescale is not a stain. It is a thermodynamic insulator bonded to the surfaces your heating system depends on.
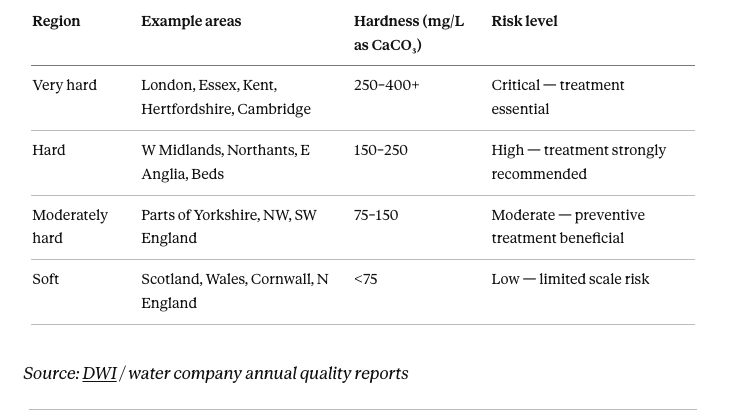
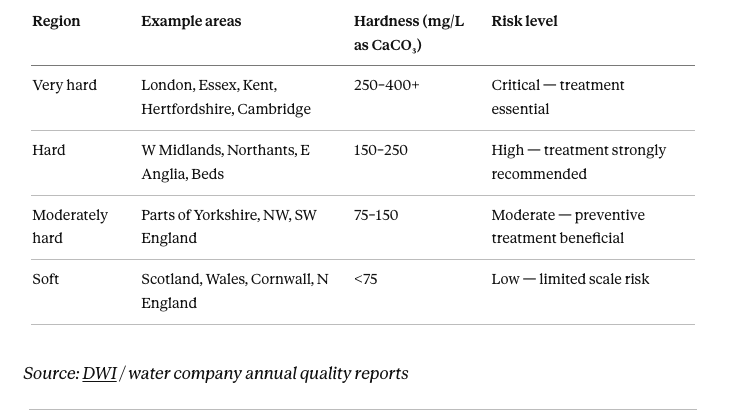
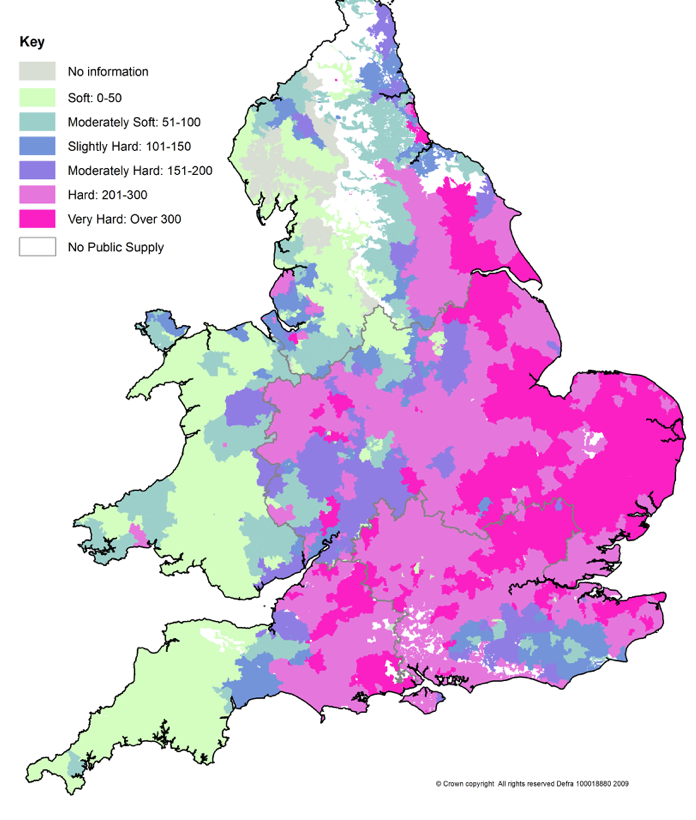
Where you are on the UK hardness map matters enormously. The Drinking Water Inspectorate publishes water quality data by postcode (dwi.defra.gov.uk) and your water company publishes annual quality reports. Check before deciding on any treatment.
UK water hardness by region:
PART 2: WHAT LIMESCALE IS ACTUALLY COSTING YOU
Most people think of limescale as a cosmetic nuisance. The financial reality is considerably more serious.
According to British Water (britishwater.co.uk), even a thin 1mm layer of limescale on heating elements can drive up energy consumption by 7–10%, adding an estimated £150–£300 to annual energy bills for a typical household. This happens because limescale acts as a thermal insulator between the heating element and the water — forcing the system to run longer and harder to reach the same temperature.
The progression over years in an untreated home:
Sources: British Water (britishwater.co.uk) and Carbon Trust (carbontrust.com)
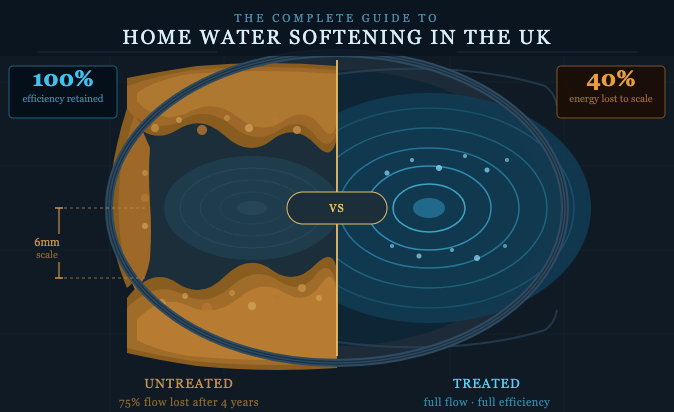
Around 6mm of limescale will build up in the average household over 4 years — causing up to a 40% reduction in your home’s energy efficiency. That is not an edge case. It is what happens in every hard water area when nothing is done.
Beyond energy bills, the costs compound: washing machines requiring up to 30% more detergent; boilers working harder and cutting their own lifespan short. Boiler replacement in the UK typically costs £2,000–£4,000. A well-maintained boiler in a treated home lasts 15–20 years; untreated, expect 8–10 years.
Check your postcode: your water company publishes hardness data free online. Search your supplier name plus “water hardness checker”, or use the DWI postcode tool at dwi.defra.gov.uk.
PART 3: THE CHEMISTRY YOU NEED TO UNDERSTAND — BICARBONATE
Here is what most water treatment guides leave out entirely: the calcium causing your limescale is only part of the story. Bicarbonate — the ion that keeps calcium dissolved and mobile in hard water — is also the primary biological buffering agent in your blood and extracellular fluid. When you strip calcium to solve the scale problem, you also strip bicarbonate. And that matters.
What bicarbonate does in water:
pH buffering: bicarbonate neutralises incoming acids by accepting hydrogen ions — the identical reaction your blood uses to resist pH shifts. Water carrying bicarbonate arrives at the gut with a stable buffering environment.
Mineral bioavailability: calcium and magnesium held in bicarbonate association remain ionically active and bioavailable. Without bicarbonate, calcium trends toward insoluble crystalline deposits that cells cannot use.
Oxidation-reduction potential (ORP): water with adequate bicarbonate has lower ORP — it is chemically less aggressive. Softened and reverse osmosis water characteristically have higher ORP — they are more oxidising and more reactive toward the surfaces they contact, including your gut lining.
What happens when bicarbonate is stripped:
The WHO review by Kozíšek (2004) — the most comprehensive scientific review of health risks from demineralised water, available at who.int — documents the consequences clearly: measurably increased renal loss of calcium, magnesium, potassium and sodium within weeks of switching to low-mineral water; continuous activation of stress hormones to compensate; and at a population level, higher rates of hypertension, ischemic heart disease, and complications in pregnancy compared with communities on adequately mineralised water.
A Swiss study found that women drinking water with both calcium and bicarbonate lost 15% less bone over 4 weeks than women on identical calcium intake but without bicarbonate — with 16% lower parathyroid hormone (PTH, the signal your body sends to pull calcium from bone to buffer acid). The buffering environment, not calcium intake alone, determines what your body does with the calcium.
Physiologically optimal drinking water targets (WHO / Kozíšek, 2004):
Calcium: 30–90 mg/L. Biological role: bone density, cardiac function, nerve signalling.
Magnesium: 17–35 mg/L. Biological role: cardiac rhythm, 300+ enzymatic reactions.
Bicarbonate: 200–250 mg/L. Biological role: pH buffering, ORP stability, cellular hydration.
Sodium: below 200 mg/L. Note: ion exchange raises sodium — relevant for hypertension patients.
Standard reverse osmosis water contains 0–10 mg/L bicarbonate. Most softened water: 30–80 mg/L. Optimal for biology: 200–250 mg/L. The gap is significant.
The critical practical point: TDS (total dissolved solids) alone is meaningless as a measure of biological water quality. Two waters at the same TDS can have bicarbonate of 40 mg/L or 120 mg/L. Always ask for alkalinity, not just hardness.
—
PART 4: HARD WATER AND YOUR HAIR AND SKIN
For many households this is the strongest immediate motivation to act — and the evidence is considerably more robust than most people realise.
Hair: calcium and magnesium ions are actively absorbed into the hair shaft, clustering around and beneath cuticle scales. This is not surface residue. Imaging studies have demonstrated calcium penetrating just beneath the cuticle, disrupting its ability to open, close and retain moisture. A 2018 study found calcium-fatty acid deposits between cuticle cells in approximately 60% of 100 donors, with high deposit loads showing dramatically lower fatigue strength and reduced shine. A 2016 study published in the Indian Journal of Dermatology (pubmed.ncbi.nlm.nih.gov/26711619/) found statistically significant magnesium deposition differences between hard and soft water groups over just three weeks.
Skin: research from the University of Sheffield and King’s College London, published in the Journal of Investigative Dermatology in 2017, identified the precise pathway: calcium and magnesium in hard water bind to surfactants in soap and shampoo, making them insoluble. These surfactant-mineral complexes precipitate onto the skin as soap scum — a thin film that cannot be rinsed away — damaging the skin barrier, increasing transepidermal water loss, and raising skin surface pH from its natural mildly acidic state toward alkaline. Alkaline pH disrupts skin barrier enzymes, leaving skin prone to bacterial colonisation and inflammation. Full study: sciencedirect.com/science/article/pii/S0022202X1732938X
Eczema: a 2021 systematic review covering 16 studies and over 385,900 participants confirmed a positive association between hard water areas (above 350 mg/L) and atopic eczema in children. One in five children and one in twelve adults in the UK suffer from eczema. Full study: onlinelibrary.wiley.com/doi/10.1111/cea.13797
The honest truth on treatment: only ion exchange fully removes calcium and magnesium from shower water. TAC conditioners reduce scale adhesion in pipes but minerals remain present in the water reaching your shower — hair and skin exposure continues. Electromagnetic devices have no peer-reviewed evidence for hair or skin improvement. Polyphosphate affects the boiler supply only and does not reach shower water at all.
—
PART 5: THE TREATMENT TECHNOLOGIES — HONEST ASSESSMENTS
Here is where one size emphatically does not fit all. Each technology has a distinct mechanism, distinct limitations, and a hardness range where it performs best.
ION EXCHANGE (TRADITIONAL SALT SOFTENER)
How it works: hard water enters a resin tank. Calcium and magnesium are captured on negatively charged resin beads, releasing sodium into the water. When the resin is exhausted, a brine flush displaces the captured minerals to drain and regenerates the resin. The output is fully softened water — no calcium, no magnesium, the familiar silky feel.
What it does well: maximum scale prevention at any hardness level. Fully eliminates mineral exposure at the shower and skin. The only clinically supported route to the soft-water feel and the most evidenced intervention for eczema management.
What it costs: installation typically £1,400–£2,200 for a quality non-electric twin-tank unit. Salt costs £100–£200 per year. Professional service every 3–5 years at £120–£185. Ten-year total cost: approximately £2,500–£4,000.
The biological trade-off: softening removes calcium, magnesium and bicarbonate from the water supply. This is not a reason not to soften — it is a reason to manage what you drink. A quality filter on an unsoftened bypass drinking tap is both legally required and biologically important (see Part 6).
Salt types: block salt (cleanest, easiest, best for most systems), tablet salt (most widely available), granular salt (cheaper but higher bridging risk), and potassium chloride (works in any salt softener — suitable for those needing to avoid sodium, but 3–4 times the cost and slightly less efficient).
TAC (TEMPLATE ASSISTED CRYSTALLISATION) CONDITIONER
How it works: TAC media converts calcium and magnesium into microscopic stable aragonite crystals that remain suspended in water and cannot adhere to pipe surfaces. Critically, the minerals are not removed. They pass through your system in a form that cannot scale, reaching your taps and your body with their ionic architecture intact. Bicarbonate is unaffected.
The evidence: DVGW-9191 certification (the most rigorous independent TAC standard) has demonstrated 99.6% scale prevention across the tested range. An Arizona State University independent evaluation (2011) found TAC achieved greater than 88% scale reduction at hardness levels of approximately 250–300 mg/L — statistically equivalent to ion exchange at that range. TAC also progressively dissolves existing scale over weeks to months, unlike ion exchange which only prevents new formation. Full study: waterboards.ca.gov/water_issues/programs/grants_loans/water_recycling/research/ion_exchange_water_softeners.pdf
What it costs: a quality tank-based TAC unit installed: £500–£900. Media replacement every five years: £100–£200. No salt, no waste water, no ongoing running cost. Ten-year total cost: approximately £700–£1,200. Near-zero maintenance.
The honest limitation: no soft-water feel. Hair and skin mineral exposure is unchanged — TAC is not a replacement for ion exchange if eczema management or the soft-water sensation is the priority.
The tipping point: below 200 mg/L, TAC is the straightforward preferred recommendation. Between 200–300 mg/L, TAC remains viable and is the default for biology-conscious households when correctly sized. Above 300 mg/L — London, SE England, parts of East Anglia — ion exchange provides greater certainty, but this is also where the biological drinking water cost is highest, making a quality bypass filter non-negotiable.
POLYPHOSPHATE DOSING
How it works: food-grade polyphosphate dosed at the boiler cold feed coats pipe surfaces, inhibiting calcium from forming scale. Minerals not removed. Approved for drinking water use. Very low cost, near-zero maintenance. Does not treat water reaching the shower or other taps.
Where it fits: boiler protection on a tight budget, or as a secondary measure alongside another treatment. Annual cartridge cost: £15–£25. Unit: £50–£100. Ten-year cost: approximately £400–£700.
ELECTROMAGNETIC AND ELECTROLYTIC DESCALERS
How they work: electromagnetic devices use pulsed EM fields to encourage calcite to crystallise as aragonite rather than the adherent calcite form. Alloy-based electrolytic devices use passive reactions to similar effect. No plumbing modification required. Minerals remain fully in water.
Honest assessment: independent testing rates electromagnetic devices at approximately half the scale prevention efficiency of TAC, and a fraction of ion exchange. Evidence at hardness levels above 200 mg/L is significantly weaker. No peer-reviewed evidence for hair or skin improvement. Their place is in rented properties or listed buildings where plumbing cannot be modified, or as a low-cost first step in moderate hardness areas (75–200 mg/L). Cost: £70–£400 depending on unit.
—
PART 6: UK LAW — WHAT YOU MUST KNOW BEFORE INSTALLING A SOFTENER
This is consistently misunderstood. When you install an ion exchange softener in the UK, a separate unsoftened cold water tap is not merely good practice — it is required by the governing regulatory framework.
The Water Supply (Water Fittings) Regulations 1999, Schedule 2, Paragraph 26 requires that all domestic premises have at least one tap providing water suitable for human consumption. The WRAS Code of Practice (endorsed by British Water) recommends a mains water tap providing unsoftened water for drinking wherever reasonably practicable. The Drinking Water Inspectorate explicitly identifies vulnerable groups — infants, people on low-sodium diets, those with hypertension or kidney disease — for whom softened water raises specific health concerns.
Key regulations:
Water Supply (Water Fittings) Regulations 1999 — legislation.gov.uk/uksi/1999/1148/schedule/2/made
WRAS Water Regulations Advisory Scheme — wras.co.uk
DWI Drinking Water Inspectorate — dwi.defra.gov.uk
Why the rule exists — the specific reasons:
Sodium: ion exchange replaces calcium and magnesium with sodium. In very hard water areas this can raise sodium to levels clinically relevant for heart failure, kidney disease and hypertension patients — and makes softened water inappropriate for infant formula preparation.
Mineral depletion: softened water is depleted in calcium, magnesium and bicarbonate. Long-term consumption is associated with measurably higher renal mineral loss and documented cardiovascular and bone effects.
Pipe metal leaching: soft water, lacking calcium and bicarbonate buffering, is chemically aggressive toward copper and lead pipes. Cases of elevated lead in infant formula mixed with soft water have been documented.
Practical compliance: a separate cold tap in the kitchen must be plumbed to the unsoftened supply, bypassing the softener. For households with infants, always use the unsoftened tap for formula preparation — the NHS and DWI position is clear on this.
Important: none of these regulations apply to TAC conditioners, electromagnetic descalers, or polyphosphate systems. They do not alter ionic composition and a separate bypass tap is not required.
—
PART 7: MATCHING TREATMENT TO YOUR HOUSEHOLD
The right treatment depends on four factors working together: water hardness, biological priorities, hair and skin needs, and budget.
Summary comparison:
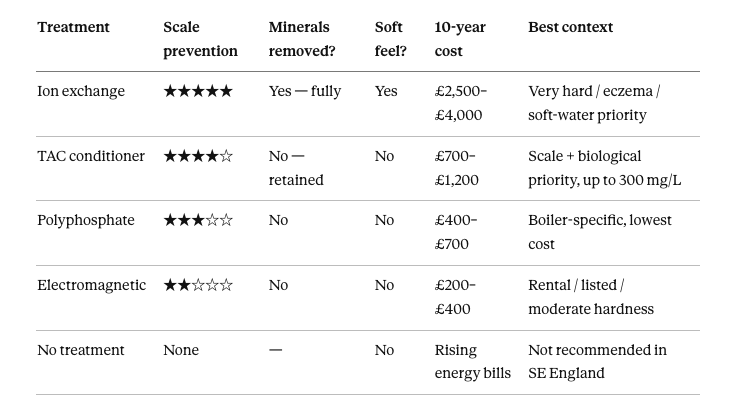
Ion exchange (salt softener): five-star scale prevention. Minerals fully removed. Gives soft-water feel. Ten-year cost approximately £2,500–£4,000. Best for: very hard water, eczema, soft-water priority.
TAC conditioner: four-star scale prevention. Minerals fully retained. No soft-water feel. Ten-year cost approximately £700–£1,200. Best for: scale prevention with biological water quality priority, up to 300 mg/L.
Polyphosphate: three-star scale prevention, boiler only. Minerals retained. Ten-year cost approximately £400–£700. Best for: boiler-specific, lowest cost entry.
Electromagnetic: two-star scale prevention. Minerals retained. Ten-year cost approximately £200–£400. Best for: rental properties, listed buildings, moderate hardness.
No treatment: no scale prevention. Ten-year cost: rising energy bills and premature appliance replacement. Not recommended in SE England.
Very hard water areas (250+ mg/L — London, SE England, Cambridge, Ipswich, Norwich): at these levels, scale damage is substantial and progressive. The town with the hardest water in the UK is Ipswich at 378 ppm; Norwich at 346 ppm and St Albans at 342 ppm follow closely. Ion exchange provides the only guaranteed full scale elimination. A quality drinking water filter on the unsoftened bypass tap is non-negotiable — legally required and biologically essential.
Hard to moderately hard (75–250 mg/L — Midlands, NW England, SW England, parts of Yorkshire): TAC is the recommended default — excellent scale prevention, full mineral retention, no ongoing consumables, biologically optimal water. If the household has eczema or wants the soft-water feel, ion exchange remains the only route.
For biology-conscious households at any hardness: test bicarbonate (alkalinity), not just hardness. Your water company’s annual report gives alkalinity by postcode free. Target drinking water: calcium 30–90 mg/L, magnesium 17–35 mg/L, bicarbonate 200–250 mg/L. If ion exchange is already installed, a quality under-sink carbon filter on the unsoftened bypass tap is the single most important addition.
—
THE EIGHT THINGS EVERY UK HOMEOWNER SHOULD KNOW
1. Limescale is not cosmetic. According to British Water, just 1.6mm of scale on a heating element increases energy use by 12%. 6mm — common after four years untreated — causes 40% efficiency loss.
2. The financial case is clear. Over ten years in a hard water area without treatment, the combined cost of excess energy, excess detergent and premature appliance replacement typically exceeds the cost of any treatment system available.
3. Bicarbonate is water’s biological architecture. Optimal for human health is 200–250 mg/L. Most softened or RO water contains less than 80 mg/L. This gap has documented health consequences at a population level.
4. Ion exchange is the gold standard for scale and soft-water feel — but removes calcium, magnesium and bicarbonate. A quality filter on the unsoftened bypass tap is legally required and biologically important.
5. TAC is the preferred default below 300 mg/L for biology-conscious households. Near-equivalent scale protection — DVGW-certified at 99.6% — without removing any minerals. No salt, no drain connection, no ongoing consumable cost. Not appropriate if eczema management or soft-water feel is the priority.
6. Hard water damages hair and skin through a specific mechanism. Only ion exchange eliminates mineral exposure at the shower.
7. A separate unsoftened cold tap is a legal requirement after ion exchange installation under the Water Supply (Water Fittings) Regulations 1999. This does not apply to TAC or electromagnetic systems.
8. TDS is not a measure of water quality. Ask for alkalinity. Two waters at the same TDS can have completely different biological value.
—
SCIENTIFIC REFERENCES AND FURTHER READING
Limescale and energy costs:
British Water — limescale efficiency data — britishwater.co.uk
Carbon Trust — energy efficiency and limescale — carbontrust.com
Parliamentary evidence on hard water and energy efficiency — committees.parliament.uk/writtenevidence/6832/html/
Bicarbonate, minerals and water biology:
WHO — Calcium and magnesium in drinking water: public health significance (2009) — who.int/publications/i/item/9789241563550
Wynn E. et al. — Nutritional alkalinity and bone mass (European Journal of Nutrition, 2009) — pubmed.ncbi.nlm.nih.gov/19241044/
Hair and skin research:
Jabbar-Lopez et al. — Water hardness and atopic eczema: systematic review (Clinical & Experimental Allergy, 2021) — onlinelibrary.wiley.com/doi/10.1111/cea.13797
Danby et al. — Effect of water hardness on surfactant deposition and skin irritation (Journal of Investigative Dermatology, 2017) — sciencedirect.com/science/article/pii/S0022202X1732938X
Srinivasan G. et al. — Effects of hard water on hair tensile strength (NIH, 2013) — pmc.ncbi.nlm.nih.gov/articles/PMC3927171/
Indian Journal of Dermatology — Hard vs soft water hair shaft changes (2016) — pubmed.ncbi.nlm.nih.gov/26711619/
Book a free water quality consultation: thewaterdr.com/consultation/
The Water Dr — Independent water treatment specialists — Bristol, South West England and Wales
Educational content only. Does not constitute medical or engineering advice. Verify water hardness and treatment suitability for your specific supply before installation.